
What are LEDs?
LEDs are a type of semiconductor called "Light Emitting Diode". White LEDs, which have achieved practical realization through the use of high-brightness blue LEDs developed in 1993 based on Gallium Nitride, are attracting increased attention as a 4th type of light source.

How do LEDs Emit Light?
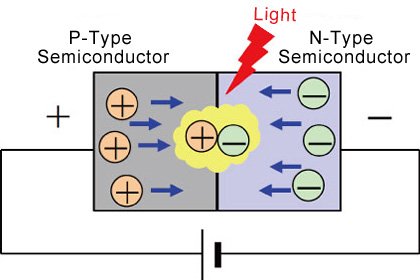
LEDs (Light Emitting Diodes) are semiconductor light sources that combine a P-type semiconductor (larger hole concentration) with an N-type semiconductor (larger electron concentration). Applying a sufficient forward voltage will cause the electrons and holes to recombine at the P-N junction, releasing energy in the form of light.
Compared with conventional light sources that first convert electrical energy into heat, and then into light, LEDs (Light Emitting Diodes) convert electrical energy directly into light, delivering efficient light generation with little-wasted electricity.
Types of LEDs
Two types of LEDs are available, a lamp type (leaded) and a chip type (surface mount). Users can select the ideal type based on set requirements.

Wavelength and Color
The LED color (emission wavelength) will change depending on materials used. This makes it possible to customize the color to meet certain wavelength specifications required for applications that use traditional bulbs as light sources (for which standards exist), such as traffic lights and automotive lamps.
Two specifications for wavelength are used to indicate color: λP (Peak Wavelength) and λD (Dominant Wavelength), with λD corresponding to the color actually seen by the human eye.
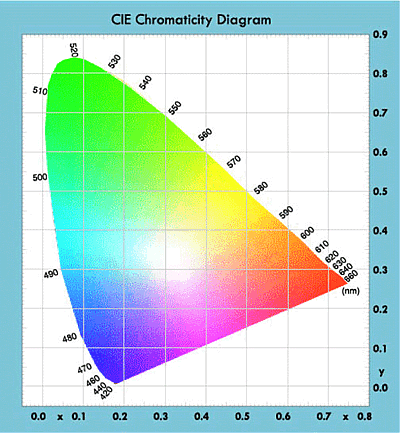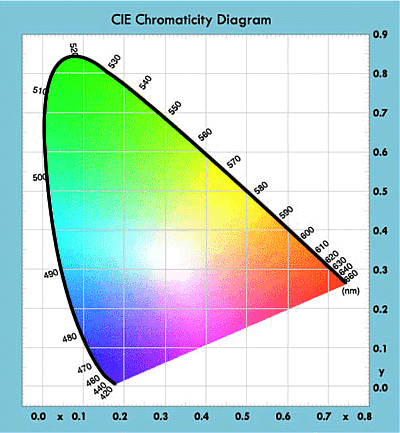
Different wavelengths involved in the process determine the different colors produced from the LEDs. Hence, light emitted by the device depends on the type of semiconductor material used.
Infrared light is produced by using Gallium Arsenide (GaAs) as a semiconductor. Red or yellow light is produced by using Gallium-Arsenide-Phosphorus (GaAsP) as a semiconductor. Red or green light is produced by using Gallium-Phosphorus (GaP) as a semiconductor.
The LED color (emission wavelength) will change depending on materials used. This makes it possible to customize the color to meet certain wavelength specifications required for applications that use traditional bulbs as light sources (for which standards exist), such as traffic lights and automotive lamps.
Two specifications for wavelength are used to indicate color: λP (Peak Wavelength) and λD (Dominant Wavelength), with λD corresponding to the color actually seen by the human eye.

Different wavelengths involved in the process determine the different colors produced from the LEDs. Hence, light emitted by the device depends on the type of semiconductor material used.
Infrared light is produced by using Gallium Arsenide (GaAs) as a semiconductor. Red or yellow light is produced by using Gallium-Arsenide-Phosphorus (GaAsP) as a semiconductor. Red or green light is produced by using Gallium-Phosphorus (GaP) as a semiconductor.

Wouldn't the PN junction run out of holes and electrons to recombine after some time?
ReplyDelete